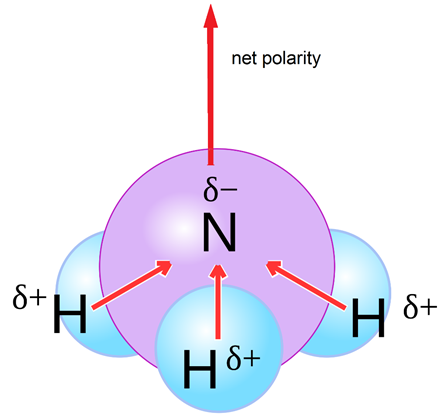
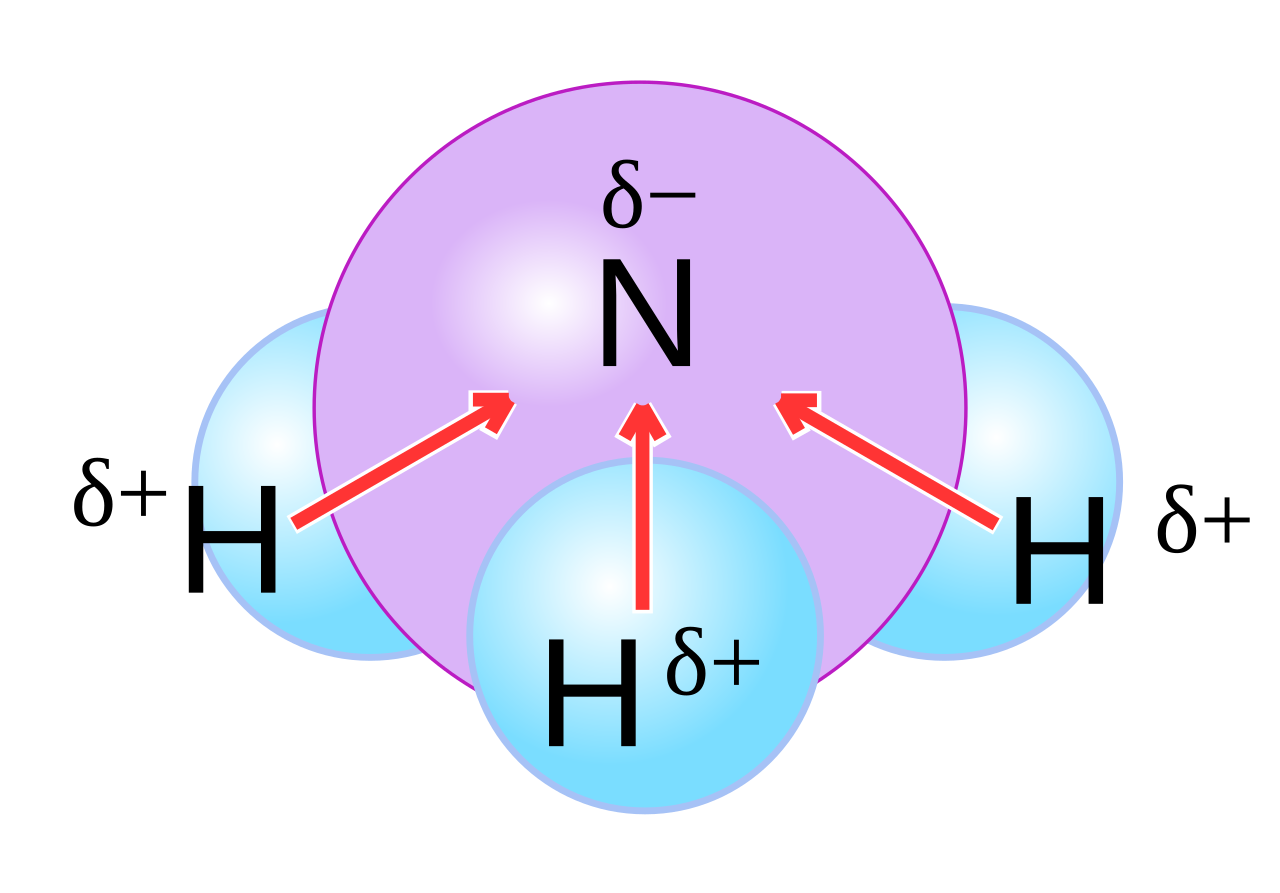
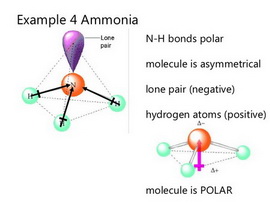
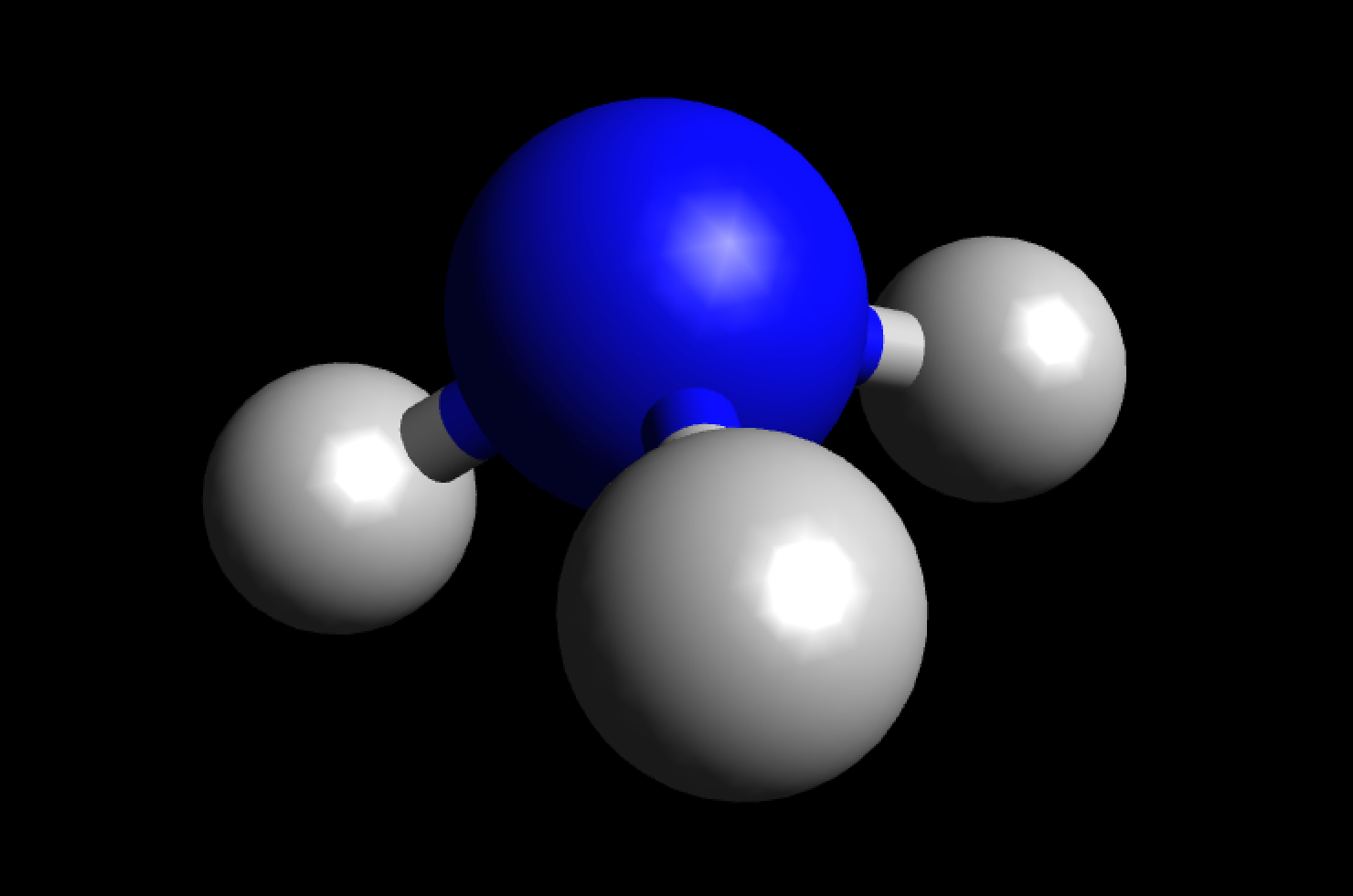
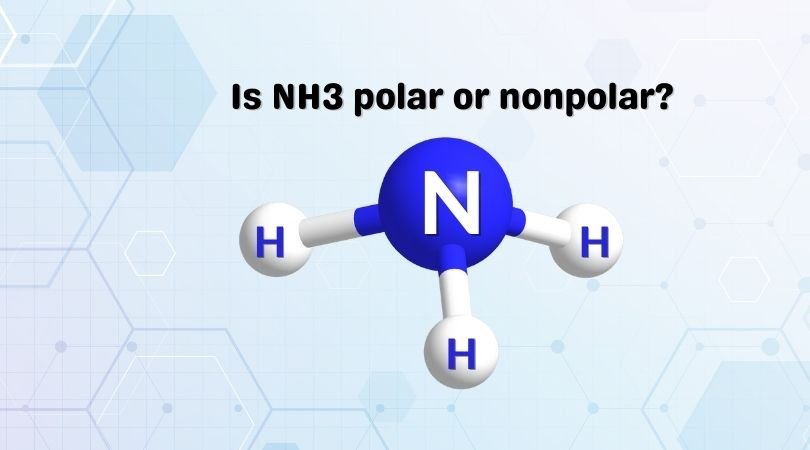
Why is ammonia (NH3) a polar molecule while boron trifluoride (BF3) is a nonpolar molecule? What is the difference in the polarity of these compounds? - Quora
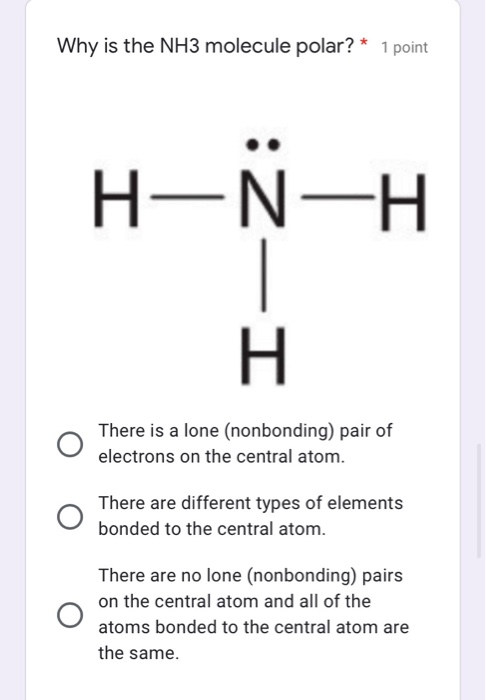
![What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C](https://www.vedantu.com/question-sets/0f8d7d3e-59fc-4e46-bb6f-9eeb5b3d71541941507909701512327.png)
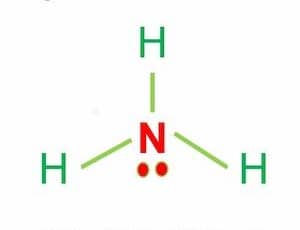
What is the bond polarity of \\[N{H_3}\\]?A) \\[N{H_3}\\] is a non-polar moleculeB) \\[N{H_3}\\] is a polar molecularC) \\[N - H\\] bond is polarD) Both B and C

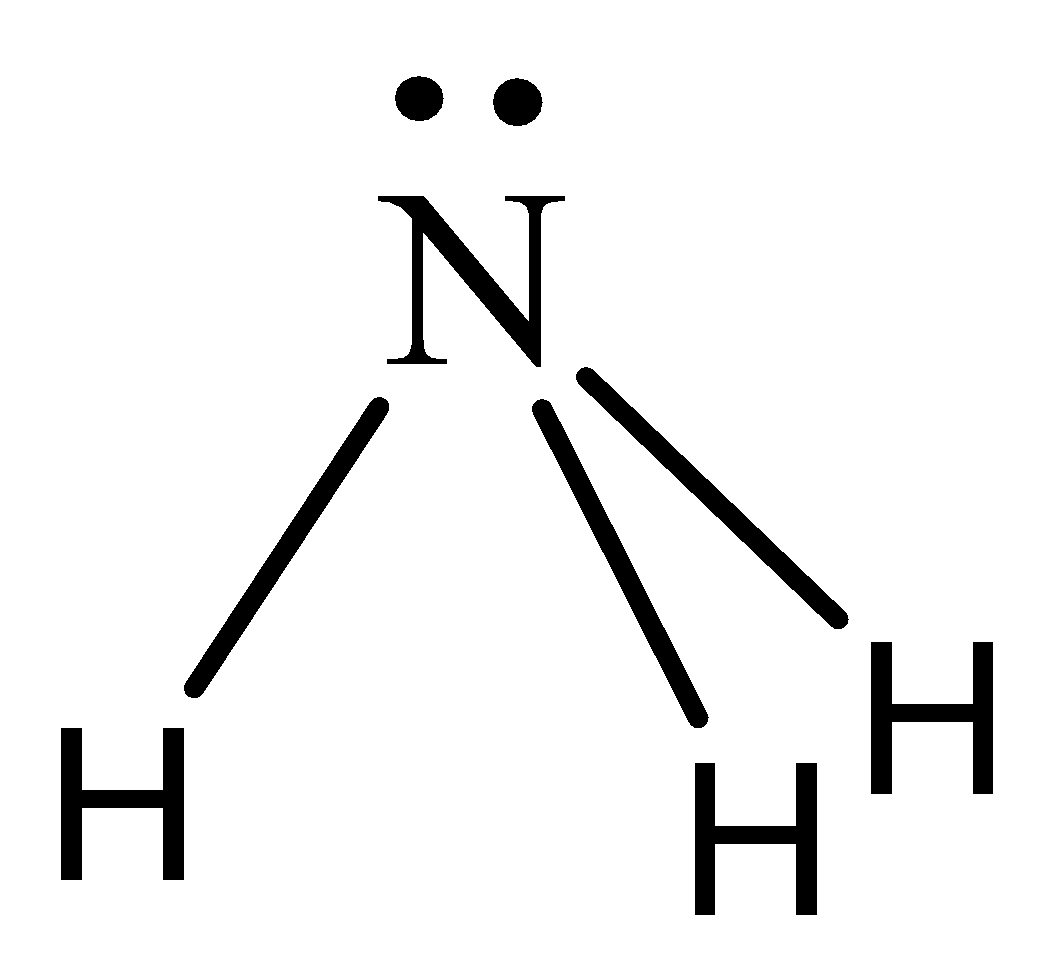
Draw the Lewis structure for NH3 and state its molecular geometry. Is it polar or nonpolar? | Homework.Study.com